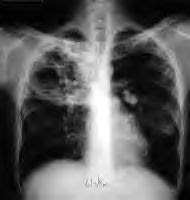
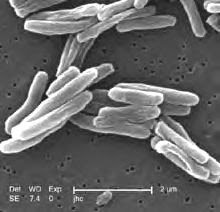
Brief Communication: Characteristics of Spontaneous Cases of Tuberculosis Associated with Infliximab Angela Raval, PharmD; Gita Akhavan-Toyserkani, PharmD, MBA; Allen Brinker, MD, MS; and Mark Avigan, MD, CM
20 November 2007 Volume 147 Issue 10 Pages 699-702
Background: A warning for tuberculosis was added to the approved labeling for infliximab in October 2001.
Objective: To describe adverse event reports of tuberculosis during infliximab therapy after labeling changes.
Design: Case series.
Setting: Spontaneous adverse event reports maintained in the Adverse Event Reporting System database in the United States.
Patients: 130 patients with infliximab-associated tuberculosis.
Measurements: Clinical and laboratory data.
Results: The U.S. Food and Drug Administration received 130 domestic, spontaneous reports of tuberculosis in patients treated with infliximab between 1 November 2001 and 30 May 2006, including 59 (45%) with extrapulmonary disease. The most commonly reported risk factors included concomitant immunosuppressant use (n = 89), history of latent or active tuberculosis (n = 33), and being born into or having spent extensive time in an area where tuberculosis is endemic (n = 25). In the subset of 67 cases with documented initiation of infliximab therapy after the drug labeling change, 34 patients with a negative tuberculin skin test result before initiation of infliximab therapy developed tuberculosis after receiving infliximab.
Limitation: Conclusions from spontaneous case reports may not be generalizable to the entire infliximab-receiving population.
Conclusion: Clinicians should be vigilant in screening and monitoring for tuberculosis in patients receiving infliximab
WHAT'S NEW IN TUBERCULOSIS
Wednesday, 21 November 2007
Monday, 12 November 2007
Aftermath report on the Speaker fiasco
Breaches in security that allowed a U.S. tuberculosis patient to defy health officials and fly to Europe and back in May can be at least partly fixed with faster communication and better training, according to a federal government report.A report from the U.S. Centers for Disease Control and Prevention on its handling of the matter shows a need for more coordination between airlines and federal agencies in such emergencies, including quick transmission of passenger information.A copy of the "After Action Report" obtained by Reuters on Thursday shows a lack of clear standard operating procedures allowed Atlanta lawyer Andrew Speaker to fly to Greece and Italy for his wedding and honeymoon against direct advice from authorities and then sneak back into the United States. .The report, which has been circulating among congressional staffers, says local officials also need to be told of new, flexible CDC powers to stop people from traveling. Local officials in Georgia said they could not legally act until Speaker had already disobeyed their orders.Better and faster tests for TB are also needed. Speaker did not learn that he had anything but ordinary TB for weeks because testing is so slow.Cetron noted that the CDC has only half the number of staff needed to properly watch travelers at ports of entry as recommended by the Institute of Medicine. A 2005 report from the Institute said CDC needed 1 inspector for every 750,000 travelers."I think there is this idea out there that somehow we will be erecting impenetrable fortresses at ports of entry that, rendered well, will leave us impervious to infectious diseases. That's just not going to happen," Cetron said in a telephone interview.
http://tinyurl.com/yufhj7
http://tinyurl.com/yufhj7
South African scientists crack drug-resistant TB code
South African scientists have sequenced the entire genome of a strain of extremely drug-resistant Mycobacterium tuberculosis (XDR-TB).
Scientists from the Nelson R. Mandela School of Medicine at the University of KwaZulu Natal, and the National Genomics Platform sequenced the genome 20 times to distinguish mutations from sequencing errors and provide a reference for further sequencing projects of XDR and multidrug-resistant TB. Lifelab, a funding mechanism of the South African Department of Science and Technology, funded the sequencing initiative. The cost of the research has not been disclosed.
James Sakwa, manager of the National Genomics Platform, told SciDev.Net that the next step will be to to develop a diagnostic kit that can quickly and efficiently diagnose this strain of XDR-TB. The breakthrough was achieved by using "pyro-sequencing" technology, where massive amounts of information are produced in parallel.
"This enabled us to sequence the whole genome within a week," he said. If the scientists had used older technologies, it would have taken about a year to achieve the same result.Proposals for the sequencing of other TB strains are currently being considered by the National Genomics Platform. "The truth is we don't know how many mutations of XDR TB there are," Sakwa said.The findings were announced at a press conference in Durban, KwaZulu Natal province, South Africa in October.
http://tinyurl.com/345l8g
Sunday, 11 November 2007
new drug, SQ109
A new tuberculosis drug given special status by both U.S. and European regulators might lead to simpler, more effective TB treatment regimens.Approximately one-third of the world’s population is infected with the tuberculosis bacterium, and approximately 1.6 million people died of the disease during 2005.Currently, TB patients must adhere to a complex treatment regimen over a six- to nine-month period. That demanding schedule, researchers said, often results in patients skipping treatment doses, which, in turn, gives rise to drug-resistant strains such as multi-drug-resistant, or MDR, tuberculosis and the recently identified extensively drug-resistant form of the disease.
The new drug, SQ109, is an antimicrobial agent developed through a partnership between the National Institute of Allergy and Infectious Diseases and the biotech company Sequella Inc.SQ109 has recently been granted "orphan drug" status by the U.S. Food and Drug Administration and the European Medicines Agency. The orphan designation involves tax reductions and marketing exclusivity to encourage development of drugs to treat diseases affecting fewer than 200,000 people.
http://tinyurl.com/yp3u6o
The new drug, SQ109, is an antimicrobial agent developed through a partnership between the National Institute of Allergy and Infectious Diseases and the biotech company Sequella Inc.SQ109 has recently been granted "orphan drug" status by the U.S. Food and Drug Administration and the European Medicines Agency. The orphan designation involves tax reductions and marketing exclusivity to encourage development of drugs to treat diseases affecting fewer than 200,000 people.
http://tinyurl.com/yp3u6o
OPEN SOCIETY INSTITUTE ON TB EPIDEMIC IN EUROPE AS INFECTION RATES
Controlling tuberculosis in Europe requires the participation of community groups and activists, according to an official “offer of partnership” to be presented to Europe’s ministers on Monday,
October 22 at the Ministerial Forum, “All Against Tuberculosis,” convened by the World Health Organization (WHO) and the German government. Ministers of health, finance, and justice from the 53 European Member States are expected to declare TB a “public health threat” and to commit new resources to tackle the epidemic in Europe.
TB is a preventable and curable disease. Yet, in 2005, 65,700 people in the European region died from TB, according to the WHO. The countries of the former Soviet Union account for 72 percent of all cases of TB reported in the European region. Greece, Sweden, and the United Kingdom also reported significant increases in TB infections in recent years.
“Rises in the rates and severities of infections make it clear that TB can no longer be dismissed as a disease of the past,” said George Soros, OSI Chairman. “I applaud the European ministers for finally recognizing TB as a regional health priority. Europe should also increase funding to fight TB in the developing world, where it continues to be one of the top killers.”
The WHO has invited Zemfira Kondur, an advocate from the Ukraine who works with marginalized Roma communities, to present civil society’s offer of partnership at the Ministerial Forum. “The burden of TB is greatest on the most marginalized communities, including Roma and other ethnic minorities,” said Kondur. “We urge our government leaders to accept our offer of partnership so we can work together to end this disease of poverty and inequality once and for all.” Most TB cases are concentrated among groups who have limited access to health care or who live in conditions that facilitate the spread of TB.
In London, TB outbreaks have disproportionately impacted homeless people, prisoners, and people who use drugs.
Cases of drug-resistant TB in Europe are among the worst in the world, and the number of people infected with both HIV and TB is also growing.
“The outbreak of HIV and TB co-infection has a devastating impact on patients, their families, and communities,” said Paul Thorn, director of the Tuberculosis Survival Project. “In order to control the spread of TB, we need health services to work with us, not just for us.”
http://tinyurl.com/29aw8s
Monday, 5 November 2007
South Africa projected increase in XDR-TB rates
Without new interventions, cases of extensively drug-resistant tuberculosis (XDR-TB) in rural South Africa will increase dramatically over the next five years, according to a study published 27 October in The Lancet. The study, which modelled the effect of various infection control measures on the spread of XDR-TB in the rural community of Tugela Ferry in KwaZulu-Natal, South Africa, suggests that infection rates will increase from 194 cases in 2007 to an estimated average of 234 cases a year by 2012. Multidrug-resistant TB will also increase from 352 cases in 2007 to 425 a year over the same period. They estimate that 72––96 per cent of all new XDR-TB cases in Tugela Ferry will occur in people infected with HIV. The study suggests that a combination of controls —— including using masks, reducing hospitalisation time, improving ventilation and rapid drug resistance testing —— could avert almost half the predicted XDR-TB cases by 2012 at Tugela Ferry and at similar resource-limited hospitals around the country.
http://tinyurl.com/2tadul
http://tinyurl.com/2tadul
Subscribe to:
Posts (Atom)